Induction of defense-related enzymes in tomato plants inoculated with selected rhizobacteria against bacterial wilt disease
Main Article Content
Abstract
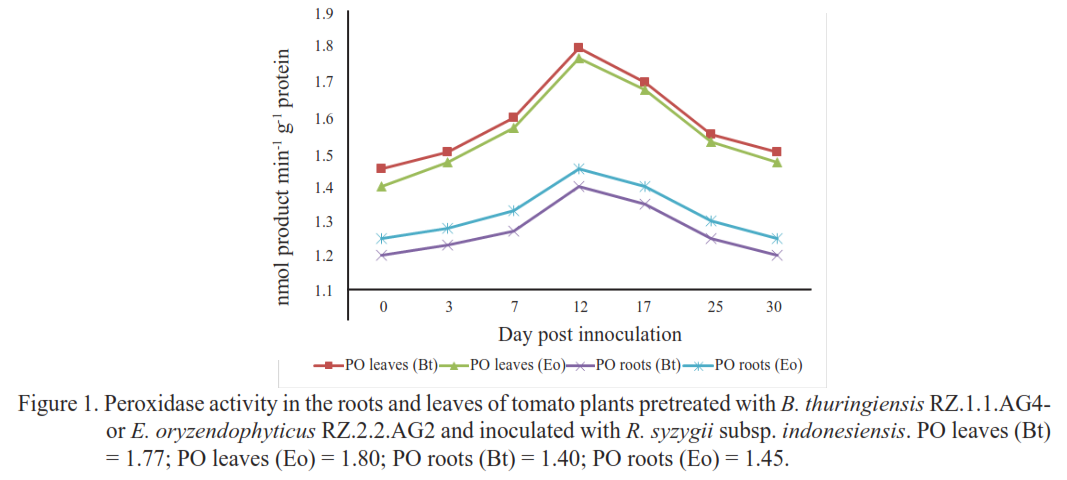
Bacterial wilt caused Ralstonia syzygii subsp. indonesiensis is one of the most destructive disease of tomato and results in severe yield losses. Plant growth-promoting rhizobacteria (PGPR) are known to enhance plant growth and health. In previous research, seven selected PGPR strains were identified for their ability to suppress R. syzygii subsp. indonesiensis and promote tomato growth. The present study aimed to evaluate the enzymatic defense responses of tomato plants inoculated with two selected rhizobacterial strains. The strains were assessed for their ability to induce the production of defense-related enzymes, including peroxidase (PO), phenylalanine ammonia-lyase (PAL), polyphenol oxidase (PPO), and lipoxygenase (LO). R. syzigii subsp. indonesiensis was inoculated onto tomato plants seven days after rhizobacterial treatment at the seedling stage. Enzyme activities were recorded from 0 to 30 days after pathogen inoculation at two-day intervals. The results showed a reduction in disease incidence and severity in tomato plants treated with the two rhizobacterial strains compared with the control. E. oryzendophyticus RZ.2.2.AG2 demonstrated greater effectiveness in reducing disease incidence (26%) and severity (2.5) than B. thuringiensis RZ.1.1.AG4. Enzyme activity assays further confirmed that both strains induced plant resistance by increasing PAL, PO, PPO, and LO activities, with significantly higher enzyme activity observed in roots than in leaves. Tomato plants treated with E. oryzendophyticus RZ.2.2.AG2 exhibited the highest enzyme activities in both roots and leaves, indicating that disease suppression was associated with induced systemic resistance (ISR) mediated through activation of defense-related enzymes.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
Akram A, Ongena M, Duby F, Dommes J, & Thonart P. 2008. Systemic resistance and lipoxygenase-related defense response induced in tomato by Pseudomonas putida strain BTP1. BMC Plant Biol. 8: 113. https://doi.org/10.1186/1471-2229-8-113
Axelrod B, Cheesbrough TM, & Laakso S. 1981. Lipoxygenases from soybean: EC 1.13.11.12 Linoleate:oxygen oxidoreductase. In: Lowenstein JM (Ed.). Methods in Enzymology. 71: 441–451. https://doi.org/10.1016/0076-6879(81)71055-3
Balamurugan A, Sakthivel K, Gautam RK, Sharma SK, & Kumar A. 2021. Ralstonia solanacearum: Biology and its management in solanaceous vegetable crops. In: Sharma SK, Singh UB, Sahu PK, Singh HV, Sharma PK (Eds.). Rhizosphere Microbes: Soil and Plant Functions. pp. 259–289. Springer. Singapore.
Bateman DF. 1967. Increase in polyphenol oxidase in diseased plant tissue. In: Kelman A (Ed.). Source Book of Laboratory Exercise in Plant Pathology. pp. 208–209. W.H. Freeman and Co. San Francisco.
Baysal T & Demirdöven A. 2007. Lipoxygenase in fruits and vegetables: A review. Enzyme Microb. Technol. 40(4): 491–495. https://doi.org/10.1016/j.enzmictec.2006.11.025
Chen C, Bélanger RR, Benhamou N, & Paulitz TC. 2000. Defense enzymes induced in cucumber roots by treatment with plant growth-promoting rhizobacteria (PGPR) and Pythium aphanidermatum. Physiol. Mol. Plant Pathol. 56(1): 13–23. https://doi.org/10.1006/pmpp.1999.0243
Constabel CP, Bergey DR, & Ryan CA. 1996. Polyphenol oxidase as a component of the inducible defense response in tomato against herbivores. In: Romeo JT, Saunders JA, Barbosa P (Eds.). Phytochemical Diversity and Redundancy in Ecological Interactions. pp 231–252. Plenum Press. New York.
De Meyer G, Capieau K, Audenaert K, Buchala A, Métraux JP, & Höfte M. 1999. Nanogram amount of salicylic acid produced by rhizobacterium Pseudomonas aeruginosa 7NSK2 activate the systemic acquired resistance pathway in bean. MPMI. 12(5): 450–458. https://doi.org/10.1094/MPMI.1999.12.5.450
Devi B, Tiwari M, Yadav N, & Singh P. 2023. Intergenerational immune priming: Harnessing plant growth promoting rhizobacteria (PGPR) for augmented wheat protection against spot blotch. Physiol. Mol. Plant Pathol. 128: 102164. https://doi.org/10.1016/j.pmpp.2023.102164
Dickerson DP, Pascholati SF, Hagerman AE, Butler LG, & Nicholson RL. 1984. Phenylalanine ammonia-lyase and hydroxylcinnamate: CoA ligase in maize mesocotyls inoculated with Helminthosporium maydis or H. carbonum. Physiol. Mol. Plant Pathol. 25(2): 111–123. https://doi.org/10.1016/0048-4059(84)90050-X
Duan C, Yu J, Bai J, Zhu Z, & Wang X. 2014. Induced defense responses in rice plants against small brown planthopper infestation. Crop J. 2(1): 55–62. https://doi.org/10.1016/j.cj.2013.12.001
Edy N, Anshary A, Lakani I, Balosi F, & Zahlin N. 2022. Rapid detection of Ralstonia syzygii subsp. syzygii on cloves vascular disease and identification potential alternate host. Asia-Pac. J. Sci. Technol. 28(2): 1–7. https://doi.org/10.14456/apst.2023.21
El-Argawy E & Adss IA. 2016. Quantitative gene expression of peroxidase, polyphenoloxidase and catalase as molecular markers for resistance against Ralstonia solanacearum. Am. J. Mol. Biol. 6(2): 88–100. https://doi.org/10.4236/ajmb.2016.62010
Feussner I & Wasternack C. 2002. The lipoxygenase pathway. Annu Rev Plant Biol. 53: 275–297. https://doi.org/10.1146/annurev.arplant.53.100301.135248
Gogoi P, Phukan T, & Saikia R. 2024. Systemic resistance induced by plant growth-promoting rhizobacteria in Bhut Jolokia (Capsicum chinense Jacq.) suppressed the collar rot disease. Sci. Hortic. 324: 112625. https://doi.org/10.1016/j.scienta.2023.112625
Gonçalves LSA, Rodrigues R, Diz MSS, Robaina RR, do Amaral Júnior AT, Carvalho AO, & Gomes VM. 2013. Peroxidase is involved in Pepper yellow mosaic virus resistance in Capsicum baccatum var. pendulum. Genet. Mol. Res. 12(2): 1411–1420. https://doi.org/10.4238/2013.April.26.3
Habazar T, Yanti Y, & Ritonga C. 2014. Formulation of indigenous rhizobacterial isolates from healthy soybean’s root, which ability to promote growth and yield of soybean. IJASEIT. 4(5): 75–79.
Habazar T, Yanti Y, Reflinaldon, Yaherwandi, Nasution CR, & Felia S. 2018. In vitro characterization of selected indigenous rhizobacterial strains as biocontrol agent of bacterial wilt disease on chili. J. Biopest. 11(1): 14–24. https://jbiopestic.com/journals/vol_11_1_14-24.pdf
Hamid S, Lone R, & Mohamed HI. 2021. Production of antibiotics from PGPR and their role in biocontrol of plant diseases. In: Mohamed HI, El-Beltagi HEDS, Abd-Elsalam KA (Eds.). Plant Growth-Promoting Microbes for Sustainable Biotic and Abiotic Stress Management. pp. 441–461. Springer International Publishing. Switzerland. https://doi.org/10.1007/978-3-030-66587-6_16
Hammerschmidt R, Nucckles F, & Kuć I. 1982. Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum lagenarium. Physiol. Plant Pathol. 20(1): 73–76. https://doi.org/10.1016/0048-4059(82)90025-X
Han YQ, Li P, Gong SL, Yang L, Wen LZ, & Hou ML. 2016. Defense responses in rice induced by silicon amendment against infestation by the leaf folder Cnaphalocrocis medinalis. PLoS One. 11(4): e0153918. https://doi.org/10.1371/journal.pone.0153918
Hildebrand M, Hallick RB, Passavant CW, & Borque DP. 1988. Trans-splicing in chloroplasts: the rps 12 loci of Nicotiana tabacum. Proc. Natl. Acad. Sci. U.S.A. 85(2): 372–376. https://doi.org/10.1073/pnas.85.2.372
Horita M & Tsuchiya K. 2001. Genetic diversity of Japanese strains of Ralstonia solanacearum. Phytopathology. 91(4): 399–407. https://doi.org/10.1094/PHYTO.2001.91.4.399
Jha Y. 2019. Higher induction of defense enzymes and cell wall reinforcement in maize by root associated bacteria for better protection against Aspergillus niger. J. Plant Prot. Res. 59(3): 341–349. https://doi.org/10.24425/jppr.2019.129746
Ji X, Lu G, Gai Y, Zheng C, & Mu Z. 2008. Biological control against bacterial wilt and colonization of mulberry by an endophytic Bacillus subtilis strain. FEMS Microbiol. Ecol. 65(3): 565–573. https://doi.org/10.1111/j.1574-6941.2008.00543.x
Jiang Y & Miles PW. 1993. Generation of H2O2 during enzymic oxidation of catechin. Phytochemistry. 33(1): 29–34. https://doi.org/10.1016/0031-9422(93)85391-4
Kandan A, Radjacommare R, Ramiah M, Ramanathan A, & Samiyappan R. 2003. PGPR induced systemic resistance in cowpea against Tomato spotted wilt virus by activating defense against tomato spotted wilt virus by activating defense related enzymes and compound. In: Sarma YR (Ed.). Proceedings of the Sixth International Workshop on Plant Growth Promoting Rhizobacteria. pp. 480–486. Calicut, India.
Kawano T. 2003. Roles of the reactive oxygen species-generating peroxidase reactions in plant defense and growth induction. Plant Cell Rep. 21(9): 829–837. https://doi.org/10.1007/s00299-003-0591-z
Khan A, Bano A, Khan RA, & Khan N. 2023. Role of PGPR in suppressing the growth of Macrophomina phaseolina by regulating antioxidant enzymes and secondary metabolites in Vigna radiata (L.) R. Wilczek. S. Afr. J. Bot. 158: 443–451. https://doi.org/10.1016/j.sajb.2023.05.040
La Camera S, Gouzerh G, Dondt S, Hoffmann L, Fritig B, Legrand M, & Heitz T. 2004. Metabolic reprogramming in plant innate immunity: the contributions of phenylpropanoid and oxylipin pathways. Immunol. Rev. 198(1): 267–284. https://doi.org/10.1111/j.0105-2896.2004.0129.x
Li L & Steffens JC. 2002. Overexpression of polyphenol oxidase in transgenic tomato plants results in enhanced bacterial disease resistance. Planta. 215(2): 239–247. https://doi.org/10.1007/s00425-002-0750-4
Maketon M, Apisitsantikul J, & Sirizweekul C. 2008. Greenhouse evaluation of Bacillus subtilis AP-01 and Trichoderma harzianum AP-001 in controlling tobacco diseases. Braz. J. Microbiol. 39(2): 296–300. https://doi.org/10.1590/s1517-838220080002000018
Mayer AM & Harel E. 1979. Polyphenol oxidases in plants. Phytochemistry. 18(2): 193–215. https://doi.org/10.1016/0031-9422(79)80057-6
Mayer AM, Harel E, & Shaul RB. 1965. Assay of catechol oxidase—a critical comparison of methods. Phytochemistry. 5(4): 783–789. https://doi.org/10.1016/S0031-9422(00)83660-2
Mayer AM. 2006. Polyphenol oxidases in plants and fungi: Going places? A review. Phytochemistry. 67(21): 2318–2331. https://doi.org/10.1016/j.phytochem.2006.08.006
Messiha NAS, van Diepeningen AD, Farag NS, Abdallah SA, Janse JD, & van Bruggen AHC. 2007. Stenotrophomonas maltophilia: A new potential biocontrol agent of Ralstonia solanacearum, causal agent of potato brown rot. Eur. J. Plant Pathol. 118(3): 211–225. https://doi.org/10.1007/s10658-007-9136-6
Mohammadi M & Kazemi H. 2002. Changes in peroxidase and polyphenol oxidase activities in susceptible and resistant wheat heads inoculated with Fusarium graminearum and induced resistance. Plant Sci. 162(4): 491–498. https://doi.org/10.1016/S0168-9452(01)00538-6
Nakkeeran S, Kavitha K, Chandrasekar G, Renukadevi P, & Fernando WGD. 2006. Induction of plant defence compounds by Pseudomonas chlororaphis PA23 and Bacillus subtilis BSCBE4 in controlling damping-off of hot pepper caused by Pythium aphanidermatum. Biocontrol Sci. Technol. 16(4): 403–416. https://doi.org/10.1080/09583150500532196
Neshat M, Abbasi A, Hosseinzadeh A, Sarikhani MR, Chavan DD, & Rasoulnia A. 2022. Plant growth promoting bacteria (PGPR) induce antioxidant tolerance against salinity stress through biochemical and physiological mechanisms. Physiol. Mol. Biol. Plants. 28(2): 347–361. https://doi.org/10.1007/s12298-022-01128-0
Nguyen MT & Ranamukhaarachchi SL. 2010. Soil-borne antagonists for biological control of bacterial wilt disease caused by Ralstonia solanacearum in tomato and pepper. J. Plant Pathol. 92(2): 395–406. https://www.jstor.org/stable/41998815
Nurdika AAH, Arwiyanto T, & Sulandari S. 2022. Physio-biochemical, molecular characterization, and phage susceptibility of Ralstonia pseudosolanacearum associated with tomato (Solanum lycopersicum) and eggplant (Solanum melongena). Biodiversitas. 23(10): 5149–5158. https://doi.org/10.13057/biodiv/d231022
Ohta H, Ida S, Mikami B, & Morita Y. 1986. Changes in lipoxygenase components of rice seedling during germination. Plant Cell Physiol. 27(5): 911–918. https://doi.org/10.1093/oxfordjournals.pcp.a077178
Ongena M, Duby F, Rossignol F, Fauconnier ML, Dommes J, & Thonart P. 2004. Stimulation of the lipoxygenase pathway is associated with systemic resistance induced in bean by a nonpathogenic Pseudomonas strain. MPMI. 17(9): 1009–1018. https://doi.org/10.1094/MPMI.2004.17.9.1009
Ongena M, Jourdan E, Adam A, Paquot M, Brans A, Joris B, Arpigny JL, & Thonart P. 2007. Surfactin and fengycin lipopeptides of Bacillus subtilis as elicitors of induced systemic resistance in plants. Environ Microbiol. 9(4): 1084–1090. https://doi.org/10.1111/j.1462-2920.2006.01202.x
Pandey S, Fartyal D, Agarwal A, Shukla T, James D, Kaul T, Negi YK, Arora S, & Reddy MK. 2017. Abiotic stress tolerance in plants: Myriad roles of ascorbate peroxidase. Front. Plant Sci. 8: 581. https://doi.org/10.3389/fpls.2017.00581
Passardi F, Penel C, & Dunand C. 2004. Performing the paradoxical: How plant peroxidases modify the cell wall. Trends Plant Sci. 9(11): 534–540. https://doi.org/10.1016/j.tplants.2004.09.002
Patel JS, Kumar G, Bajpai R, Teli B, Rashid M, & Sarma BK. 2021. PGPR formulations and application in the management of pulse crop health. In: Rakshit A, Meena VS, Parihar M, Singh HB, Singh AK (Eds.). Biofertilizers: Volume 1: Advances in Bio-Inoculants. pp. 239–251. Woodhead Publishing. https://doi.org/10.1016/B978-0-12-821667-5.00012-9
Pereira LF, Goodwin PH, & Erickson L. 2000. Peroxidase activity during susceptible and resistant interactions between cassava (Manihot esculenta) and Xanthomonas axonopodis pv. manihotis and Xanthomonas cassavae. J. Phytopathol. 148(11–12): 575–578. https://doi.org/10.1111/j.1439-0434.2000.00548.x
Podile AR & Laxmi VDV. 1998. Seed bacterization with B. subtilis AF1 increases Phenylalanine Ammonia-lyase and reduces the incidence of Fusarial wilt in pigeonpea. J Phytopathol. 146(5–6): 255–259. https://doi.org/10.1111/j.1439-0434.1998.tb04687.x
Raj SN, Sarosh BR, & Shetty HS. 2006. Induction and accumulation of polyphenol oxidase activities as implicated in development of resistance against pearl millet downy mildew disease. Funct. Plant Biol. 33(6): 563–571. https://doi.org/10.1071/FP06003
Rajinimala P, Rabindran R, Ramaiah M, Nagarajan P, & Varanavasiappan S. 2003. PGPR mediated disease resistance in bitter gourd against Bitter gourd yellow mosaic virus. In: Proceedings of the 6th international PGPR workshop. pp. 5–10. Calicut, India.
Ramamoorthy V, Raguchander T, & Samiyappan R. 2002. Induction of defense-related proteins in tomato roots treated with Pseudomonas fluorescens Pf1 and Fusarium oxysporum f.sp. lycopersici. Plant Soil. 239(1): 55–68. https://doi.org/10.1023/A:1014904815352
Resti Z, Habazar T, Putra DP, & Nasrun. 2016. Peroxidase Enzyme Activity of The Introduced Shallots with Endophytic Bacteria and Resistant to Bacterial Leaf Blight (Xanthomonasaxonopodis pv. allii). J Trop Plant Pest Dis.16(2): 131–137. https://doi.org/10.23960/j.hptt.216131-137
Reuveni R, Shimoni M, Karchi Z, & Kuc J. 1992. Peroxidase activity as a biocontrol marker for resistance of muskmelon (Cucumis melo) to Pseudoperonospora cubensis. Phytopathology. 82(7): 749–753. https://doi.org/10.1094/Phyto-82-749
Sailaja PR, Podile AR, & Reddanna P. 1998. Biocontrol strain of Bacillus subtilis AF1 rapidly induces lipoxygenase in groundnut (Arachis hypogaea L.) compared to crown rot pathogen Aspergillus niger. Eur. J. Plant Pathol. 104(2): 125–132. https://doi.org/10.1023/A:1008638608639
Salazar B, Ortiz A, Keswani C, Minkina T, Mandzhieva S, Singh SP, Rekadwad B, Borriss R, Jain A, Singh HB, & Sansinenea E. 2023. Bacillus spp. as bio-factories for antifungal secondary metabolites: Innovation beyond whole organism formulations. Microb. Ecol. 86(1): 1–24. https://doi.org/10.1007/s00248-022-02044-2
Sarvanan T, Bhaskaran R, & Muthuswamy M. 2004. Pseudomonas fluorescens induced enzymological changes in banana roots (cv. Rasthali) against Fusarium wilt disease. Plant Pathol. J. 3(2): 72–80. https://doi.org/10.3923/ppj.2004.72.80
Seleim MAA, Saead FA, Abd-El-Moneem KMH, & Abo-Elyousr KAM. 2011. Biological control of bacterial wilt of tomato by plant growth promoting rhizobacteria. Plant Pathol. J. 10(4): 146–153. https://doi.org/10.3923/ppj.2011.146.153
Seth K, Vyas P, Deora S, Gupta AK, Meena M, Swapnil P, & Harish. 2023. Understanding plant-plant growth-promoting rhizobacteria (PGPR) interactions for inducing plant defense. In: Swapnil P, Meena M, Harish, Marwal A, Vijayalakshmi S, & Zehra A (Eds.). Plant-Microbe Interaction-Recent Advances in Molecular and Biochemical Approaches. pp. 201–226. Academic Press. London. https://doi.org/10.1016/B978-0-323-91876-3.00010-5
Shah J. 2005. Lipids, lipases, and lipid-modifying enzymes in plant disease resistance. Annu Rev Phytopathol. 43(1): 229–260. https://doi.org/10.1146/annurev.phyto.43.040204.135951
Shi C, Dai Y, Xia B, Xu X, Xie Y, & Liu Q. 2001. The purification and spectral properties of polyphenol oxidase I from Nicotiana tabacum. Plant Mol. Biol. Rep. 19: 381–382. https://doi.org/ 10.1007/BF02772840
Silva HSA, Romeiro RdS, Macagnan D, Halfeld-Vieira BdA, Pereira MCB, & Mounteer A. 2004. Rhizobacterial induction of systemic resistance in tomato plants: Non-specific protection and increase in enzyme activities. Biol. Control. 29(2): 288–295. https://doi.org/10.1016/S1049-9644(03)00163-4
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, & Klenk DC. 1985. Measurement of protein using bicinchoninic acid. Anal. Biochem. 150(1): 76–85. https://doi.org/10.1016/0003-2697(85)90442-7
Stevens LH, Davelaar E, Kolb RM, Pennings EJM, & Smit NPM. 1998. Tyrosine and cysteine are substrates for blackspot synthesis in potato. Phytochemistry. 49(3): 703–707. https://doi.org/10.1016/S0031-9422(98)00207-6
Tahsili J, Sharifi M, Safaie N, Esmaeilzadeh-Bahabadi S, & Behmanesh M. 2014. Induction of lignans and phenolic compounds in cell culture of Linum album by culture filtrate of Fusarium graminearum. J. Plant Interact. 9(1): 412–417. https://doi.org/10.1080/17429145.2013.846419
Thakker JN, Patel S, & Dhandhukia PC. 2013. Induction of defense-related enzymes in banana plants: Effect of live and dead pathogenic strain of Fusarium oxysporum f.sp. cubense. Int. Sch. Res. Notices. 2013(1): 601303. https://doi.org/10.5402/2013/601303
Van Loon LC. 1998. Induced resistance in plants and the role of pathogenesis-related proteins. Eur J. Plant Pathol. 103(9): 753–765. https://doi.org/10.1023/A:1008638109140
van Peer R, Niemann GJ, & Schippers B. 1991. Induced resistance and phytoalexin accumulation in biological control of fusarium wilt of carnation by Pseudomonas sp. strain WCS417r. Phytopathology. 81(7): 726–734.
Vieira AA, De Almeida Oliveira MG, José IC, Piovesan ND, De Rezende ST, Moreira MA, & De Barros EG. 2001. Biochemical evaluation of lipoxygenase pathway of soybean plants submitted to wounding. Rev. Bras. Fisiol. Veg. 13(1): 5–12. https://doi.org/10.1590/S0103-31312001000100001
Yang P, Yuan P, Liu W, Zhao Z, Bernier MC, Zhang C, Adhikari A, Opiyo SO, Zhao L, Banks F, & Xia Y. 2024. Plant growth promotion and plant disease suppression induced by Bacillus amyloliquefaciens strain GD4a. Plants. 13(5): 672. https://doi.org/10.3390/plants13050672
Yanti Y, Astuti FF, Habazar T, & Nasution CR. 2017. Screening of rhizobacteria from rhizosphere of healthy chili to control bacterial wilt disease and to promote growth and yield of chili. Biodiversitas. 18(1): 1–9. https://doi.org/10.13057/biodiv/d180101
Yanti Y. 2015. Peroxidase enzyme activity of rhizobacteria-introduced shallots bulbs to induce resistance of shallot towards bacterial leaf blight (Xanthomonas axonopodis pv. allii). Procedia Chem. 14: 501–507. https://doi.org/10.1016/j.proche.2015.03.067
Zdor RE & Anderson AJ. 1992. Influence of root colonizing bacteria on the defense responses in bean. Plant Soil. 140(1): 99–107. https://doi.org/10.1007/BF00012811
Zhu L, Huang J, Lu X, & Zhou C. 2022. Development of plant systemic resistance by beneficial rhizobacteria: Recognition, initiation, elicitation and regulation. Front. Plant Sci. 13: 952397. https://doi.org/10.3389/fpls.2022.952397