Enhanced nematicidal potential of MnCl2-fortified Bacillus sp. cell-free supernatant against Meloidogyne incognita
Main Article Content
Abstract
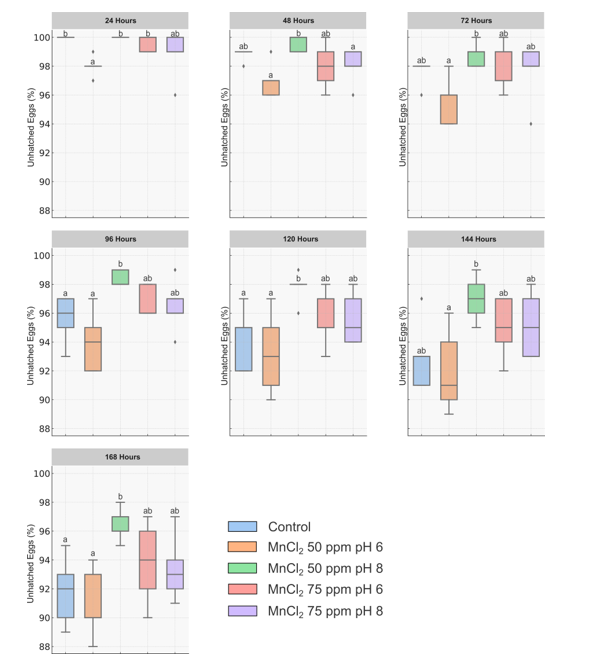
Meloidogyne incognita causes major yield losses, underscoring the need for sustainable control strategies. This study is the first to demonstrate that micronutrient fortification and pH modulation of Bacillus sp. SK07 cultures enhance the nematicidal activity of their cell-free supernatant (CFS) and alter metabolite composition. Bacillus sp. SK07 was cultured in Nutrient Broth supplemented with MnCl2 at 0, 50, or 75 ppm, with pH adjusted to 6 or 8. A 10% (v/v) CFS was evaluated in vitro against eggs and second-stage juveniles (J2) over 7 days using a completely randomized design with five treatments and five replicates. The most effective treatment—50 ppm MnCl2 at pH 8—resulted in 96.4% egg-hatch inhibition and 63.6% J2 mortality at 168 hours, significantly higher than the unfortified control (91.8% and 49.4%, respectively). Chemical analysis revealed substantial changes in metabolite composition: unfortified cultures exhibited 9 peaks, whereas the optimized treatment produced 27 peaks, indicating increased chemical diversity. Several bioactive compounds, including fatty acid derivatives and aromatic esters, were identified. These compounds belong to diverse chemical classes such as acids, esters, alcohols, and hydrocarbons, many of which are known for their nematicidal activity. These findings demonstrate that MnCl2 fortification at pH 8 effectively enhanced the bioactivity of Bacillus sp. SK07, offering a promising and sustainable approach for managing M. incognita.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
Abdel Hafez NA, Elsayed MA, El‐Shahawi MM, Awad GE, & Ali KA. 2018. Synthesis and antimicrobial activity of new thiazolidine‐based heterocycles as rhodanine analogues. J. Heterocycl. Chem. 55(3): 685–691. https://doi.org/10.1002/jhet.3087
Adiwena M, Murtilaksono A, Egra S, Hoesain M, Asyiah IN, Pradana AP, & Izatika ZN. 2023. The effects of micronutrient-enriched media on the efficacy of Bacillus subtilis as biological control agent against Meloidogyne incognita. Biodiversitas J. Biol. Divers. 24(1): 33–39. https://doi.org/10.13057/biodiv/d240105
Ahmad G, Khan A, Khan AA, Ali A, & Mohhamad HI. 2021. Biological control: A novel strategy for the control of the plant parasitic nematodes. Antonie Van Leeuwenhoek. 114(7): 885–912. https://doi.org/10.1007/s10482-021-01577-9
Asyiah IN, Mudakir I, Budiman A, & Pradana AP. 2025. Media optimization for nematode-trapping fungus Orbilia jesu-laurae and its effect in managing Meloidogyne incognita. Cogent Food Agric. 11(1): 2442667. https://doi.org/10.1080/23311932.2024.2442667
Asyiah IN, Mudakir I, Hoesain M, Pradana AP, Djunaidy A, & Sari RF. 2020. Consortium of endophytic bacteria and rhizobacteria effectively suppresses the population of Pratylenchus coffeae and promotes the growth of Robusta coffee. Biodiversitas. 21(10): 4702–4708. https://doi.org/10.13057/biodiv/d211032
Asyiah IN, Prihatin J, Hastuti AD, Winarso S, Widjayanthi L, Nugroho D, Firmansyah K, & Pradana AP. 2021. Cost-effective bacteria-based bionematicide formula to control the root-knot nematode Meloidogyne spp. on tomato plants. Biodiversitas. 22(6): 3256–3264. https://doi.org/10.13057/biodiv/d220630
Azlay L, El Boukhari MEM, Mayad EH, & Barakate M. 2023. Biological management of root-knot nematodes (Meloidogyne spp.): A review. Org. Agric. 13(1): 99–117. https://doi.org/10.1007/s13165-022-00417-y
Baatout S, Leys N, Hendrickx L, Dams A, & Mergeay M. 2007. Physiological changes induced in bacteria following pH stress as a model for space research. Acta Astronaut. 60(4–7): 451–459. https://doi.org/10.1016/j.actaastro.2006.09.012
Balogun OO, Ugoh SC, & Oladosu PO. 2022. Antimicrobial activity and GC-MS based analysis of the extract of Bacillus subtilis subsp. subtilis 168 isolated from a river bank. Innovations in Microbiology and Biotechnology. 7: 126–145. https://doi.org/10.9734/bpi/imb/v7/3649A
Bosma EF, Rau MH, van Gijtenbeek LA, & Siedler S. 2021. Regulation and distinct physiological roles of manganese in bacteria. FEMS Microbiol. Rev. 45(6): fuab028. https://doi.org/10.1093/femsre/fuab028
Cao H, Jiao Y, Yin N, Li Y, Ling J, Mao Z, Yang Y, & Xie B. 2019. Analysis of the activity and biological control efficacy of the Bacillus subtilis strain Bs-1 against Meloidogyne incognita. Crop Prot. 122: 125–135. https://doi.org/10.1016/j.cropro.2019.04.021
Desaeger J, Wram C, & Zasada I. 2020. New reduced-risk agricultural nematicides-rationale and review. J. Nematol. 52(1): e2020–2091. https://doi.org/10.21307/jofnem-2020-091
Ebone LA, Kovaleski M, & Deuner CC. 2019. Nematicides: History, mode, and mechanism action. Plant Sci. Today. 6(2): 91–97. https://doi.org/10.14719/pst.2019.6.2.468
Engelbrecht G, Horak I, Jansen van Rensburg PJ, & Claassens S. 2018. Bacillus-based bionematicides: Development, modes of action and commercialisation. Biocontrol Sci. Technol. 28(7): 629–653. https://doi.org/10.1080/09583157.2018.1469000
Gowda MT, Meena BR, Krishnan N, Manjunath M, Sellaperumal C, Rai A, Singh A, Manimurugan C, Patil J, Pandey KK, & Singh J. 2022. Antimicrobial peptides producing native Bacillus spp. for the management of root-knot nematode Meloidogyne incognita infecting okra (Abelmoschus esculentus L. Moench). Biol. Control. 171: 104951. https://doi.org/10.1016/j.biocontrol.2022.104951
Hussain T, Khan AA, & Mohamed HI. 2023. Metabolites composition of Bacillus subtilis HussainT-AMU determined by LC-MS and their effect on Fusarium dry rot of potato seed tuber. Phyton–Int. J. Exp. Bot. 92(3): 783–799. https://doi.org/10.32604/phyton.2022.026045
Jamal Q, Monkhung S, Munir S, Cho JY, Moon JH, Khattak BU, Malik MS, Younis F, & Kim KY. 2019. Identification of cyclo (L-Pro-D-Tyr) from Bacillus amyloliquefaciens Y1 exhibiting antifungal activity against Fusarium graminearum to control crown rot in wheat. Appl. Ecol. Environ. Res. 17(3): 6299–6314. https://doi.org/10.15666/aeer/1703_62996314
Karačić V, Miljaković D, Marinković J, Ignjatov M, Milošević D, Tamindžić G, & Ivanović M. 2024. Bacillus species: Excellent biocontrol agents against tomato diseases. Microorganisms. 12(3): 457. https://doi.org/10.3390/microorganisms12030457
Khan MR & Mohiddin FA. 2023. Biocontrol strategies for nematode management, an overview. In: Khan MR (Ed.). Novel Biological and Biotechnological Applications in Plant Nematode Management. pp.113–131. Springer. Singapore. https://doi.org/10.1007/978-981-99-2893-4_5
Khanna K, Kohli SK, Ohri P, Bhardwaj R, Al-Huqail AA, Siddiqui MH, Alosaimi GF, & Ahmad P. 2019. Microbial fortification improved photosynthetic efficiency and secondary metabolism in Lycopersicon esculentum plants under Cd stress. Biomolecules. 9(10): 581. https://doi.org/10.3390/biom9100581
Lahlali R, Ezrari S, Radouane N, Kenfaoui J, Esmaeel Q, El Hamss H, Belabess Z, & Barka EA. 2022. Biological control of plant pathogens: A global perspective. Microorganisms. 10(3): 596. https://doi.org/10.3390/microorganisms10030596
Li GH & Zhang KQ. 2023. Natural nematicidal metabolites and advances in their biocontrol capacity on plant parasitic nematodes. Nat. Prod. Rep. 40(3): 646–675. https://doi.org/10.1039/D2NP00074A
Liu H, Wang Y, Yang Q, Zhao W, Cui L, Wang B, Zhang L, Cheng H, Song S, & Zhang L. 2019. Genomics and LC-MS reveal diverse active secondary metabolites in Bacillus amyloliquefaciens WS-8. J. Microbiol. Biotechnol. 30(3): 417–426. https://doi.org/10.4014/jmb.1906.06055
Lu Q, Liu T, Wang N, Dou Z, Wang K, & Zuo Y. 2020. Nematicidal effect of methyl palmitate and methyl stearate against Meloidogyne incognita in bananas. J. Agric. Food Chem. 68(24): 6502–6510. https://doi.org/10.1021/acs.jafc.0c00218
Maulidia V, Soesanto L, Syamsuddin, Khairan, Hamaguchi T, Hasegawa K, & Sriwati R. 2020b. Secondary metabolites produced by endophytic bacteria against the Root-Knot Nematode (Meloidogyne sp.). Biodiversitas. 21(11): 5270–5275. https://doi.org/10.13057/biodiv/d211130
Mols M & Abee T. 2011. Primary and secondary oxidative stress in Bacillus. Environ. Microbiol. 13(6): 1387–1394. https://doi.org/10.1111/j.1462-2920.2011.02433.x
Nabilah N, Swibawa IG, Suharjo R, & Fitriana Y. 2021. Diversity and abundance of nematodes in guava (Psidium guajava L.) cultivation in Lampung. J. Trop. Plant Pests Dis. 21(2): 134–143. https://doi.org/10.23960/jhptt.221134-143
Nadeem H, Niazi P, Asif M, Kaskavalci G, & Ahmad F. 2021. Bacterial strains integrated with surfactin molecules of Bacillus subtilis MTCC441 enrich nematocidal activity against Meloidogyne incognita. Plant Biol. 23(6): 1027–1036. https://doi.org/10.1111/plb.13301
Nur MJ, Suprama, & Munif A. 2016. Keefektifan tanaman limbah Brassicaceae sebagai pengendali nematoda puru akar (Meloidogyne spp.) pada skala mikroplot di lapangan [Effectivenes of Brassicaceae plant wastes to control the root knot nematodes (Meloidogyne spp.) at a field microplot scale]. J. Trop. Plant Pests Dis. 16(2): 99–106. https://doi.org/10.23960/j.hptt.21699-106
Ntalli N, Menkissoglu-Spiroudi U, Doitsinis K, Kalomoiris M, Papadakis EN, Boutsis G, Domou M, & Monokrousos N. 2020. Mode of action and ecotoxicity of hexanoic and acetic acids on Meloidogyne javanica. J. Pest Sci. 93(2): 867–877. https://doi.org/10.1007/s10340-020-01193-y
Pandit MA, Kumar J, Gulati S, Bhandari N, Mehta P, Katyal R, Rawat CD, Mishra V, & Kaur J. 2022. Major biological control strategies for plant pathogens. Pathogens. 11(2): 273. https://doi.org/10.3390/pathogens11020273
Park MK, Lee S, & Kim YS. 2022. Effects of pH and osmotic changes on the metabolic expressions of Bacillus subtilis strain 168 in metabolite pathways including leucine metabolism. Metabolites. 12(2): 112. https://doi.org/10.3390/metabo12020112
Pradana AP, Sholehah S, Andriyani DR, Hoesain M, Astuti W, Hadiani RU, Masnilah R, Adiwena M, & Putri D. 2025. Nematicidal activity of Trichoderma harzianum-derived secondary metabolites against Meloidogyne incognita and metabolomic profiling of selected potent isolates. Asian J. Agric. 9(1): 326–338. https://doi.org/10.13057/asianjagric/g090134
Prihatiningsih N, Asnani A, & Djatmiko HA. 2021. Extracellular protease from Bacillus subtilis B315 with antagonistic activity against bacterial wilt pathogen (Ralstonia solanacearum) of chili. Biodiversitas. 22(3): 1291–1295. https://doi.org/10.13057/biodiv/d220327
Ratzke C & Gore J. 2018. Modifying and reacting to the environmental pH can drive bacterial interactions. PLOS Biol. 16(3): e2004248. https://doi.org/10.1371/journal.pbio.2004248
Saiyam D, Dubey A, Malla MA, & Kumar A. 2024. Lipopeptides from Bacillus: Unveiling biotechnological prospects—Sources, properties, and diverse applications. Braz. J. Microbiol. 55(1): 281–295. https://doi.org/10.1007/s42770-023-01228-3
Sikandar A, Zhang MY, Wang YY, Zhu XF, Liu XY, Fan HY, Xuan YH, Chen LJ, & Duan YX. 2020. Meloidogyne incognita (root-knot nematode) a risk to agriculture. Appl. Ecol. Environ. Res. 18(1): 1679–1690. https://doi.org/10.15666/aeer/1801_16791690
Singh P & Siddiqui ZA. 2010. Biocontrol of root-knot nematode Meloidogyne incognita by the isolates of Bacillus on tomato. Arch. Phytopathol. Plant Prot. 43(6): 552–561. https://doi.org/10.1080/03235400801939904
Subedi S, Thapa B, & Shrestha J. 2020. Root-knot nematode (Meloidogyne incognita) and its management: A review. J. Agric. Nat. Resour. 3(2): 21–31. https://doi.org/10.3126/janr.v3i2.32298
Swibawa IG, Fitriana Y, Solikhin S, Fiandani A, Suharjo R, Purnomo, & Susilo FX. 2024. Effectiveness of bionematicide from Purpureocillium lilacinum in controlling root-knot nematodes (Meloidogyne spp.). J. Trop. Plant Pests Dis. 24(2): 181–189. https://doi.org/10.23960/jhptt.224181-189
Tapia-Vázquez I, Montoya-Martínez AC, De los Santos-Villalobos S, Ek-Ramos MJ, Montesinos-Matías R, & Martínez-Anaya C. 2022. Root-knot nematodes (Meloidogyne spp.) a threat to agriculture in Mexico: Biology, current control strategies, and perspectives. World J. Microbiol. Biotechnol. 38(2): 26. https://doi.org/10.1007/s11274-021-03211-2
Terefe M, Tefera T, & Sakhuja P. 2012. Biocontrol (formulation of Bacillus firmus (BioNem)) of root-knot nematode, Meloidogyne incognita on tomato plants in the field. Ethiop. J. Agric. Sci. 22(1): 102–116.
Thakur S, Rana A, Sharma A, Yangchan J, Choudhary K, Kumar R, Sharma KA, Kumar S, & Sharma D. 2024. Plant nematode interaction and omics: A focus on Meloidogyne incognita. J. Crop Health. 76(6): 1281-1291. https://doi.org/10.1007/s10343-024-01025-4
Théatre A, Hoste ACR, Rigolet A, Benneceur I, Bechet M, Ongena M, Deleu M, & Jacques P. 2022. Bacillus sp.: A remarkable source of bioactive lipopeptides. In: Hausmann R & Henkel M (Eds.). Advances in Biochemical Engineering/Biotechnology. Vol 181. pp. 123–180. Springer, Cham. https://doi.org/10.1007/10_2021_182
Themuhi M, Shanthi A, Meena KS, Rajeshwaran B, Swarnakumari N, & Das D. 2025. Nematicidal investigation of crude extracts of macro basidiomycetous fungi against root-knot nematode, Meloidogyne incognita through in vitro, in vivo and in silico approaches. J. Plant Dis. Prot. 132(4): 134. https://doi.org/10.1007/s41348-025-01132-y
Tiwari S. 2024. Impact of nematicides on plant-parasitic nematodes: Challenges and environmental safety. Tunis. J. Plant Prot. 19(2): 0039. https://doi.org/10.4314/tjpp.v19i2.4
Tsai BY. 2008. Effect of temperature on the survival of Meloidogyne incognita. Plant Pathol. Bull. 17: 203–208.
Vasantha-Srinivasan P, Park KB, Kim KY, Jung WJ, & Han YS. 2025. The role of Bacillus species in the management of plant-parasitic nematodes. Front. Microbiol. 15: 1510036. https://doi.org/10.3389/fmicb.2024.1510036
Villarreal-Delgado MF, Villa-Rodríguez ED, Cira-Chávez LA, Estrada-Alvarado MI, Parra-Cota FI, & Santos-Villalobos Sdl. 2018. The genus Bacillus as a biological control agent and its implications in the agricultural biosecurity. Rev. mex. fitopatol. 36(1): 95–130. https://doi.org/10.18781/r.mex.fit.1706-5
Wang Y & Zhang Q. 2024. Improved methodology for the efficient isolation of viable Meloidogyne incognita eggs. J. Plant Dis. Prot. 131(6): 2255–2258. https://doi.org/10.1007/s41348-024-00993-z
Winarto, Yanti Y, Hamid H, & Yaherwandi. 2024 The endophytic potential, Bacillus spp. for controlling Meloidogyne sp. and increasing tomato growth and production. J. Trop. Plant Pests Dis. 24(1): 66–74. https://doi.org/10.23960/jhptt.12466-74
Xiong J, Zhou Q, Luo H, Xia L, Li L, Sun M, & Yu Z. 2015. Systemic nematicidal activity and biocontrol efficacy of Bacillus firmus against the root-knot nematode Meloidogyne incognita. World J. Microbiol. Biotechnol. 31(4): 661–667. https://doi.org/10.1007/s11274-015-1820-7
Ye L, Wang J-Y, Liu X-F, Guan Q, Dou N-X, Li J, Zhang Q, Gao Y-M, Wang M, Li J-S, & Zhou B. 2022. Nematicidal activity of volatile organic compounds produced by Bacillus altitudinis AMCC 1040 against Meloidogyne incognita. Arch. Microbiol. 204(8): 521. https://doi.org/10.1007/s00203-022-03024-3
Zhai Y, Shao Z, Cai M, Zheng L, Li G, Yu Z, & Zhang J. 2019. Cyclo (l-Pro–l-Leu) of Pseudomonas putida MCCC 1A00316 isolated from antarctic soil: Identification and characterization of activity against Meloidogyne incognita. Molecules. 24(4): 768. https://doi.org/10.3390/molecules24040768
Zhang Wp, Ruan Wb, Deng Yy, & Gao Yb. 2012. Potential antagonistic effects of nine natural fatty acids against Meloidogyne incognita. J. Agric. Food Chem. 60(46): 11631–11637. https://doi.org/10.1021/jf3036885
Zuber P. 2009. Management of oxidative stress in Bacillus. Annu. Rev. Microbiol. 63(1): 575–597. https://doi.org/10.1146/annurev.micro.091208.073241